Sizing up the problem with assessment
- Australian Society Dermal Clinicians

- Mar 5, 2021
- 5 min read
Updated: Mar 2, 2024
Early detection and identifying people at risk of developing lymphedema is very important in preventing late onset symptoms and morbidity associated with the condition. The development of secondary lymphoedema can be multifactorial, however there is developing theories that there is an underlying predisposition to lymphatic dysfunction. Secondary lymphoedema aetiology is diverse and multifactorial. For this reason patients at risk of developing lymphedema will be encountered in a wide variety of clinical settings (primary, secondary and tertiary health care providers). These include dermatology, vascular surgery, oncology, plastic surgery, and wound care. The dermal clinician will be found in all of these settings and therefore should have a thorough understanding of risk assessment, early identification of lymphatic dysfunction and referral pathways (Lymphoedema framework, 2006).

The Burden
Management of oedema is a heavy burden on the sufferer including financial cost, time and emotional investment. As such all of these considerations can affect “compliance” with therapy (Dart, 2012). To achieve optimal and maintainable outcomes for our patients we must ensure that we keep the patient at the centre of their care planning. This revolves around accurate assessment and classification to determine the most efficient and effective therapies.
50% of chronic oedema sufferers report long-term pain in oedematous limbs, which can restrict movement and cause distress.
Patients with chronic oedema are at high risk of skin breakdown and infections such as cellulitis as repeated trauma and loss of skin integrity allow opportunistic pathogens to enter.
33% of chronic oedema sufferers will develop cellulitis, 27% of these cases will require hospitalisation for IV antibiotics
The condition can be so debilitating impact sufferers psychologically that 80% of of those with lymphoedema report taking time off work, and 2% giving up employment entirely (Todd, 2013).
Risk Factors and Early Identification
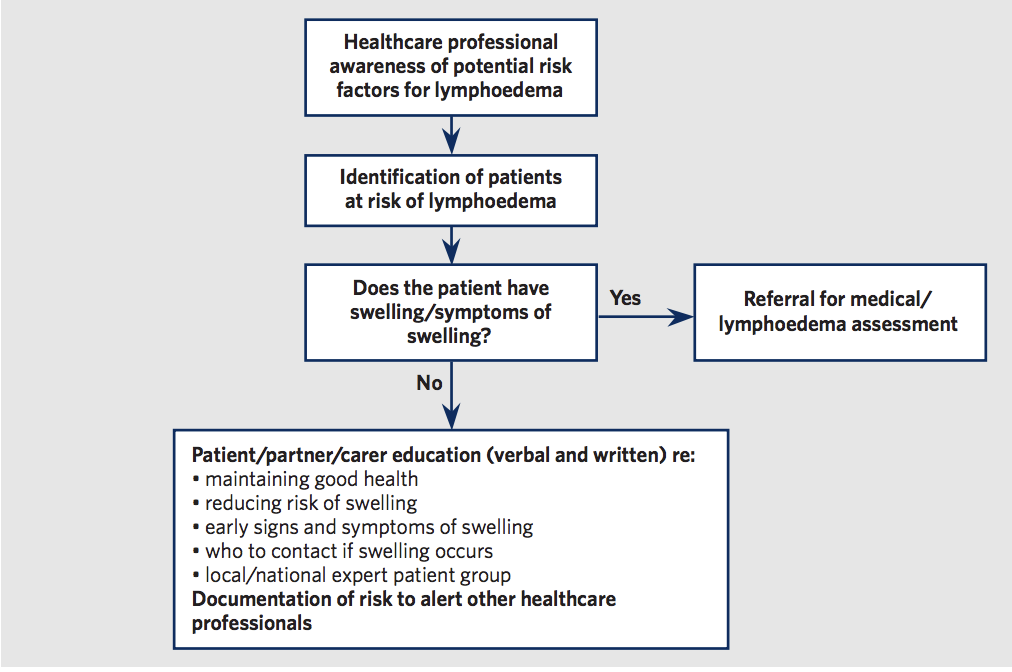
Below are Box 4, Table 1 and Figure 1 from the international consenus document on the best practice management of lymphoedema (International Lymphoedema Framework,2006). These figures summarise the most common causes or risk factors for development of chronic oedema and lymphoedema. Figure 1 presents a flow chart that is useful for the Dermal Clinician in understanding their role in identification of chronic oedema and lymphoedema. This includes the referral of those identified with chronic oedema and lymphoedma to a specialised service for diagnosis. It also outlines the importance of education that can be provided by the Dermal Clinician to those at risk in order to prevent or identify early signs of oedema so that it can be managed quickly to reduce morbidity.
Assessment: What are the early signs?
The first signs of lymphatic dysfunction and chronic oedema (subclinical signs) are usually perceivable by the patient themselves. For this reason it is important to educate clients at greater risk of what the early signs can be. Clients at greater risk include those with chronic inflammatory disorders, post surgical patients and those on medications that induce oedema.
Clothing or jewellery that is becoming tighter
Aching, pain
Feeling of heaviness, fullness, tightness or woodeness
Problems with range of movement
Obvious swelling
Those identified with chronic oedema and suspected Lymphoedema may be referred to a GP with knowledge of lymphatic dysfunction, or a specialised Lymphoedema service. At the present time there is reported to be a shortage of medical professionals and services for those with lymphatic issues. Currently patient support groups and advocacy organisations are campaigning for funding to increase hospital and outpatient services as well as cover the cost of management with a medical (medicare) rebate. At the present time there is great inconsistency in these services and funding offered nationally.
The tests below may be carried out by medical professionals or specialists. They provide information on the underlying aetiology of the oedema and provide insight into best management strategies.
Blood tests
Urine testing
Bioimpedence
X-rays
Ultrasound imaging
CT/MRI Scans
Lymphoscintography/ indocyanine green & near infrared imaging
Genetic testing
Clinical Assessment
Clinical assessment, grading and classification of Chronic Oedema and Lymphoedema allows for these conditions to be managed more effectively.
Various tools are used from the clinically useful but more subjective assessment techniques including: circumference measurements, goblet (pitting) sign and stemmer test to more objective research tools including water displacement, perometry and tonometry. Detailed skin assessment is also vital as well as assessment of nutritional status, pain, vascular function (heart, arterial and venous), mobility and psychosocial support or needs.
Watch below as to how to perform a pitting test and observe for Stemmer Sign.
Resources for Dermal Clinicians
For more information stay up and become an ASDC Member.
Other Resources can be found at:
References
Dart. L. (2012). Non-compliance? It's easy for you to say. Journal of Lymphoedema. 7(1), 8-10
Haesler. E. (2015). Evidence summary: Lymphoedema: Objective assessment using circumference measurement. Wound Practice and Research. 23(1), 36-38
Haesler. E. (2015). Evidence summary: Lymphoedema: Objective assessment using tonometry. Wound Practice and Research. 23(3), 150-151
Jones. J. (2014). Lymphoedema and chronic oedema: What’s the difference? Nursing and residential care. 16(2), 684-886
Kamble. R., Shetty. R., Diwakar. N. & Madhusudan. G. (2011). Technical note: MRI lymphangiography of the lower limb in secondary lymphedema. Indian Journal of Radiology and Imaging. 21(1),15-17
Lymphoedema Framework. (2006). Best Practice for the Management of lymphedema. International consensus. London. MEP Ltd.
Mellor. R., Bush. N., Stanton. A., Bamber. J., Levick. R., & Mortimer. P. (2004). Dual- frequency ultrasound examination of skin and subcutis thickness in breast cancer related lymphedema. The Breast Journal. 10(6), 496-503
Mendoza. E. (2013). Differential diagnosis of leg oedemas of venous and lymphatic origin. Phlebologie. 42, 153-157
Mihara. M., Hara. H., Narushima. M., Todokoro. T., Iida. T., Ohtsu. H., Murai. N., Koshima. I. (2013). Indocyanine green is superior to lymphoscintigraphy in imaging diagnosis of secondary lymphedema of the lower limbs. Journal of Vascular Surgery. 1, 194-201
Naouri. M., Samimi. M., Atlan. M., Perrodeau. E., Vallin. C., Zakine. G., Vaillant. L. & Machet. L. (2010). High resolution cutaneous ultrasonography to differentiate lipoedema from lymphedema. British Journal of Dermatology. 163, 296-301
Nikitenko. L., Shimosawa. T., Henderson. S., Makinen. T., Shimosawa. H., Qureshi. U. Pedley. B., Rees. M., Fujita. T. & Boshoff. C. (2013). Adrenomedullin Haploinsufficiency predisposes to secondary lymphedema. Journal of Investigative Dermatology. 133, 1768-1776
Piller. N. (2007). To measure or not to measure? What and when is the question. Journal of Lymphoedema. 2(2), 39-45
Todd. M. (2013). Chronic oedema: impact and management. British Journal of
Nursing. 22(11). 623-627 Tomczak. H., Nyka. W. & Lass. P. (2005). Lymphoedema: Lymphoscintigrapy versus
other diagnositic techniques: a clinician’s point of view. Nuclear Medicine
Review. 8(1), 37-43 Trayes. K., Studdiford. J., Pickle. S., & Tully. A. (2013). Edema: Diagnosis and
management. American Academy of Family Physicians. 88(2). 102-110
Varricchi. G., Yamamoto. T., Matsuda. N., Doi. K., Oshima. A., Yoshimatsu. H., Todokoro. T.
Ogata. F., Mihara. M., Narushima. M., Iida. T., & Koshima. I. (2011). The earliest finding of Indocyanine Green Lymphography in asymptomatic limbs of lower extremity lymphedema patients secondary to cancer treatment: The modified dermal backflow stage and concept of subclinical lymphedema. Plastic and Reconstructive Surgery. 127(5), 1979-1986